The Problem
Current tools for characterizing biological barriers are mismatched to the physics they need to capture. Bulk rheology averages over millimeter volumes, collapsing the spatial heterogeneity that determines transport selectivity into a single number. Particle-tracking microrheology samples only the accessible pores. Static modulus matching at a single frequency ignores the dynamic relaxation behavior that governs whether a molecule escapes a transient cage or remains trapped.
The result is an observer’s illusion: materials that are fundamentally heterogeneous appear uniform, and synthetic models that match on bulk properties fail to predict drug transport.
Our Approach
Barrier Cartography is our research program for measuring, modeling, and predicting molecular transport through biological barriers — at the resolution where the physics actually operates.
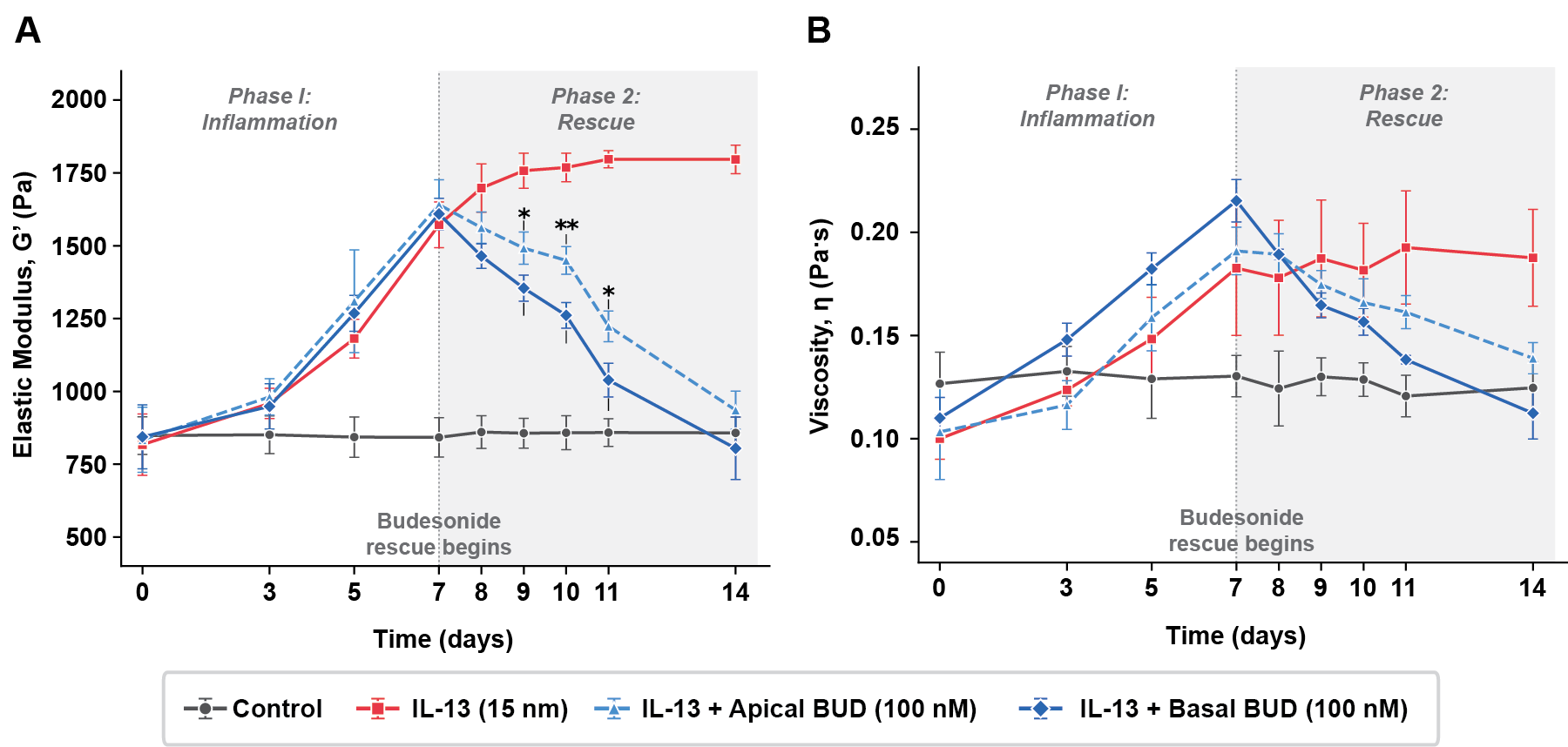
Non-contact mechanical characterization. We use resonant acoustic rheometry (RAR) to measure viscoelastic properties of mucus on living air-liquid interface cultures without contacting or disturbing the sample. This enables longitudinal monitoring of mucus mechanics under pharmacological intervention — tracking how CFTR modulators, hypertonic saline, or inflammatory stimulation change barrier properties over hours and days. Post-perturbation recovery experiments directly measure the network rearrangement time, a critical parameter that standard assays cannot capture.
Timescale competition framework. We are developing a framework that evaluates synthetic mucus models not by whether they match bulk modulus, but by whether they reproduce the transport-relevant dynamics of native mucus. Two networks with identical elastic modulus can produce fundamentally different transport if their relaxation spectra differ. Our framework identifies which timescale competitions — network relaxation vs. particle diffusion, network relaxation vs. ciliary forcing — govern outcomes, and tests whether a model faithfully reproduces them.
Toward voxel-scale mapping. The next generation of this program aims to convert fluorescence lifetime and anisotropy measurements into calibrated, spatially resolved mechanical maps of barrier environments — enabling direct visualization of where drugs are trapped, where they flow, and why.
Who Works on This
This project involves Nishant Shah (novel instrumentation and wave-based transport probes), Rahela Zaman (mucus transport and infection susceptibility), and Grace Xia (mucus viscoelasticity in nasal delivery).