The Problem
Synergistic antibiotic combinations are discovered under idealized conditions: both drugs present simultaneously, at fixed concentrations, in a homogeneous liquid. In the infected lung, none of these assumptions hold. Drugs arrive at different rates, are cleared on different timescales, and must traverse spatially heterogeneous barriers — mucus, biofilm matrix, necrotic tissue — that selectively filter based on physicochemical identity.
The result is spatiotemporal decoupling: two drugs that are synergistic in a test tube may never achieve the overlapping concentrations, at the same place, at the same time, that synergy requires. The combination fails not because the mechanism is wrong, but because the delivery environment uncouples it.
Our Approach
We reframe antimicrobial synergy as a system property — not just a drug-pair attribute — that can be preserved or broken by the transport environment. Our approach has three components:
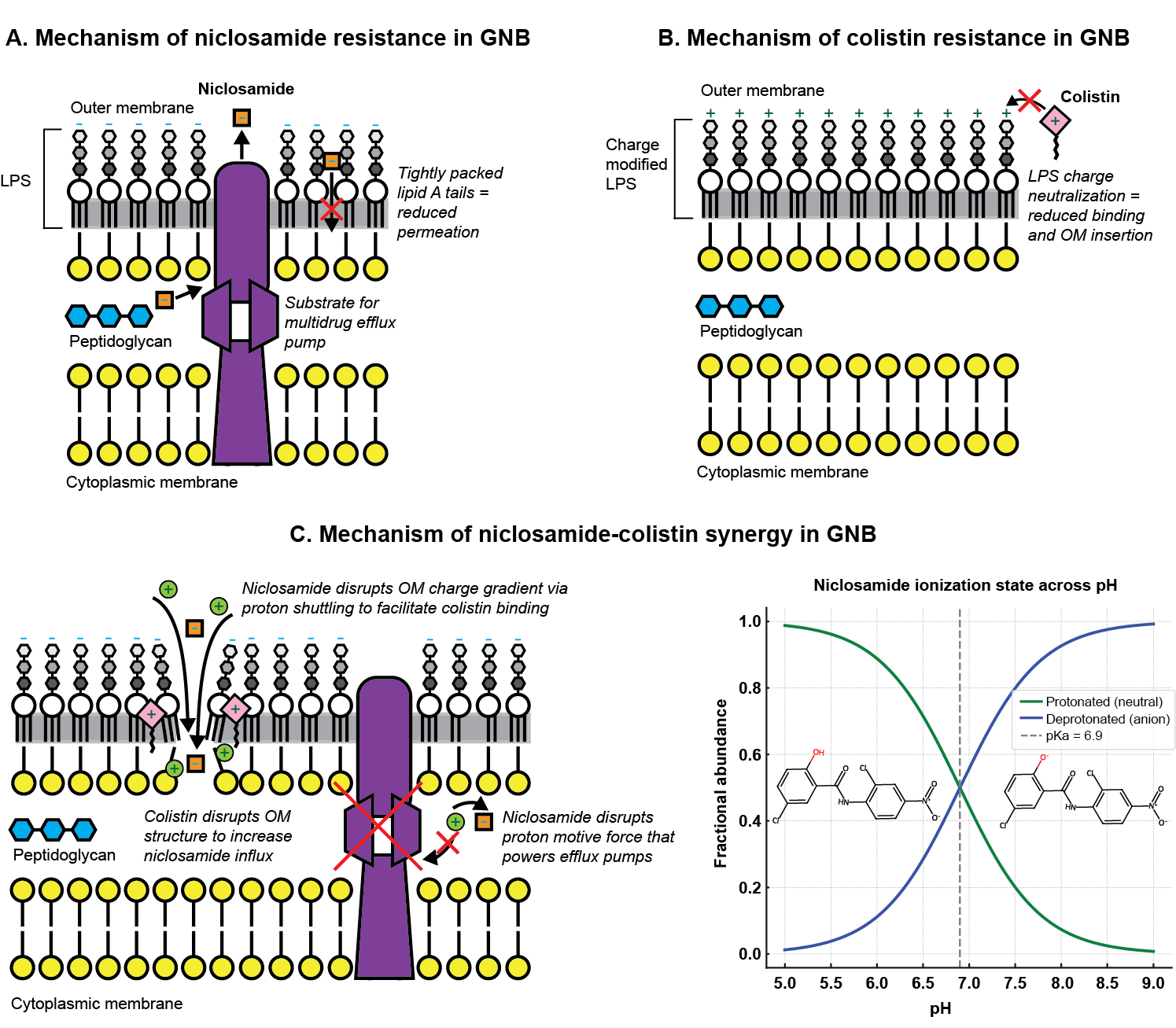
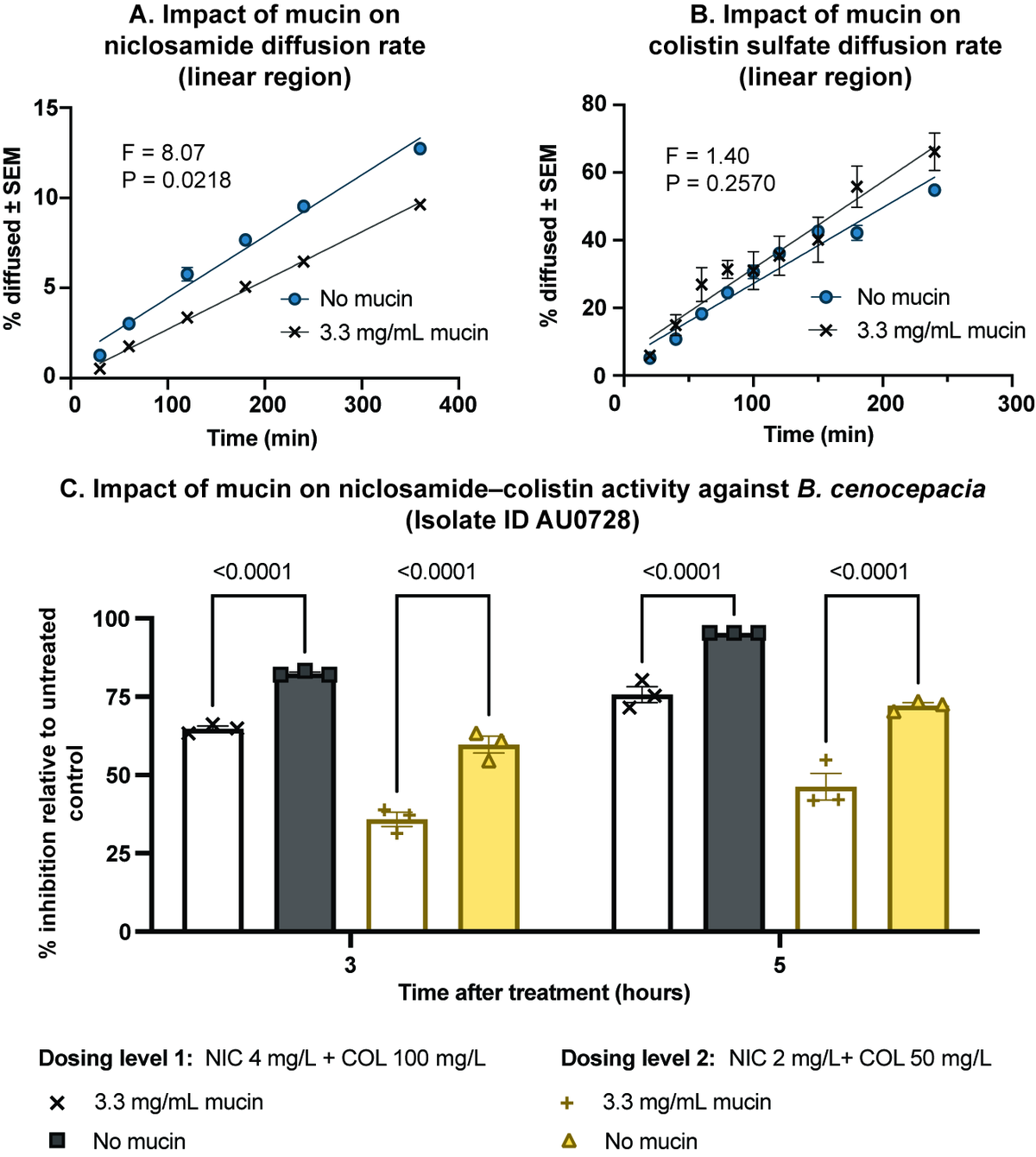
Mapping the co-exposure window. Using physiologically informed in vitro platforms that incorporate realistic mucus barriers, dynamic drug clearance, and spatial heterogeneity, we define the concentration–time window within which synergy is maintained. We showed that for niclosamide–colistin, mucin selectively impairs niclosamide diffusion while colistin passes freely, creating a spatial mismatch that abolishes synergy even when both drugs are administered simultaneously. Removing niclosamide after just two hours — mimicking its rapid pulmonary clearance — eliminated the combination’s advantage entirely.
Engineering particles that enforce co-transport. Once we know what the co-exposure window requires, we design formulations that deliver it. We developed a strategy where colistin serves a dual role: it is both the synergistic antibiotic partner and the surface-active excipient that stabilizes niclosamide microcrystals during antisolvent precipitation. The resulting spray-dried composite particles co-localize both drugs in a single respirable particle — crystalline niclosamide cores embedded in an amorphous colistin shell — achieving excipient-free, high-drug-loading inhalation powders.
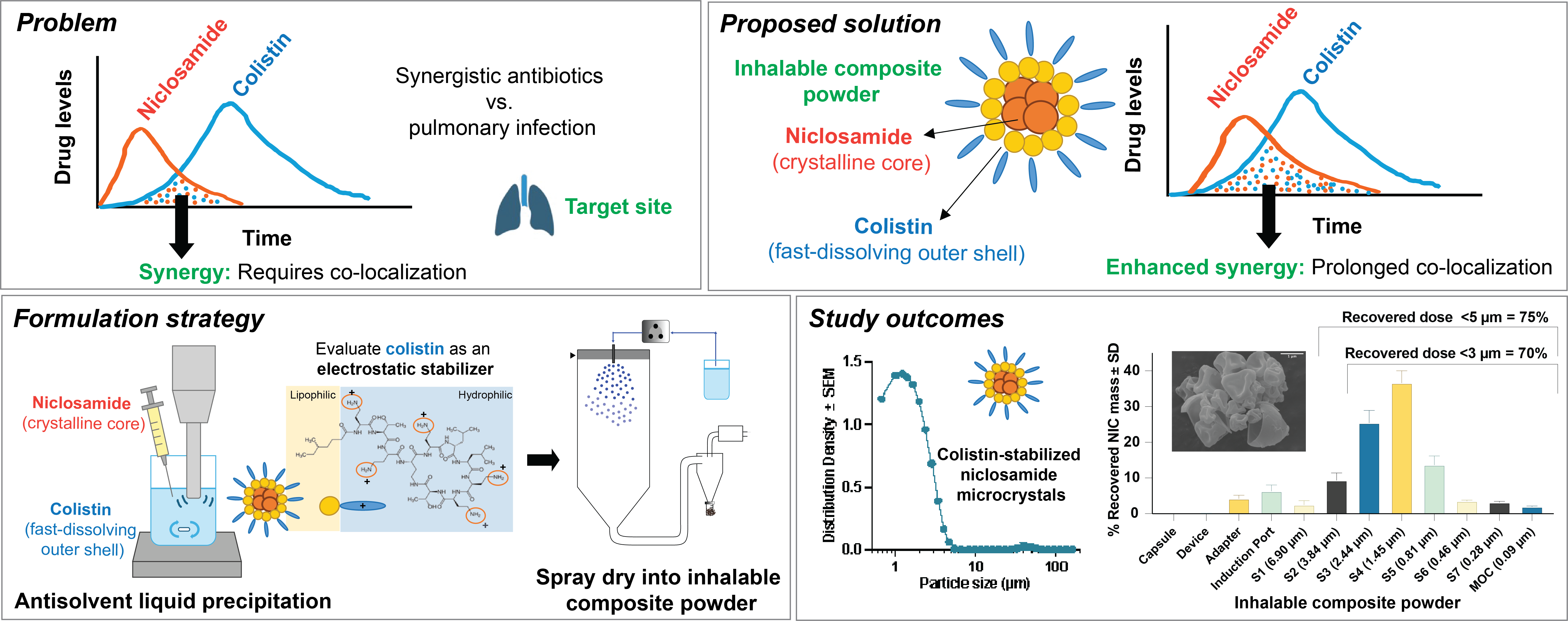

Validating in physiologically relevant models. In a murine lung infection model, intratracheal delivery of the co-processed particles reduced pulmonary bacterial burden to levels comparable to systemic ceftazidime — demonstrating that the formulation preserves therapeutic activity when delivered to the lung.
Who Works on This
This project involves Mariana Romero-Gonzalez (formulation engineering, in vitro synergy platforms), Nuz Dechayont (polymer stabilization, membrane transport), and Yice Zhang (infection models, ion-pairing strategies).
Selected Publications
- Romero-Gonzalez M et al. Colistin-stabilized antisolvent precipitation enables engineering of microcrystalline niclosamide for inhalable composite powders. International Journal of Pharmaceutics, 2026.
- Romero-Gonzalez M et al. Physiologically informed in vitro framework reveals context-dependent combinatory activity of niclosamide–colistin against Gram-negative bacteria. Journal of Antimicrobial Chemotherapy, 80(11): 2958–2969, 2025.